|
Spectrophotometry instance Current period: 0:00 Total duration: 13:06 0 energy points Science Chemistry collection Kinetics Spectrophotometry Spectrophotometry introduction Google Class room Facebook Twitter Email Spectrophotometry Spectrophotometry introduction This will be the presently selected item.
Serial Dilution Lab Purpose Movie Transcript WhatSpectrophotometry example Movie transcript What I desire to perform in this movie.
Serial Dilution Lab Purpose Free Of ChargeSpectrophotometry example Up Next Spectrophotometry example Our objective is usually to provide a free of charge, world-class education to anyone, anyplace. Usually this will be completed by evapourating or boiling, supposing that the temperature of cooking food does not affect the solute. What is definitely Chemistry Introduction Some Fundamental Definitions Hormone balance as a Research Chapter 2. Measurements Intro to Measurements Expressing Figures Significant Figures Converting Devices Other Models: Temperatures and Thickness Expressing Units End-of-Chapter Material Chapter 3. Atoms, Molecules, and Ions Intro to Atoms, MoIecules, and lons Acids Ions ánd Ionic Substances Public of Atoms and Molecules Molecules and Chemical Nomenclature Atomic Concept End-of-Chapter Materials Chapter 4. Chemical Responses and Equations Intro to Chemical substance Responses and Equations The Chemical Equation Forms of Chemical Reactions: Solitary- and Double-Displacement Responses Ionic Equations: A Closer Look Structure, Decomposition, and Combustion Responses Oxidation-Reduction Reactions Neutralization Responses End-of-Chapter Material Part 5. Stoichiometry and the Mole Introduction to Stoichiometry ánd the Mole Stoichiométry The Mole MoIe-Mass and Máss-Mass Computations Reducing Reagents The Mole in Chemical Reactions Yields End-of-Chapter Material Chapter 6. Gases Introduction to Gases Pressure Gasoline Laws and regulations Other Gas Laws and regulations The Ideal Gas Regulation and Some Applications Gas Blends Kinetic Molecular Concept of Gases Molecular Effusion and Diffusion Real Gases End-of-Chapter Material Part 7. 
Electronic Construction Launch to Electronic Framework Firm of EIectrons in Atoms EIectronic Framework and the Periodic Desk Quantum Amounts for Electrons Light Periodic Tendencies End-of-Chapter Material Section 9. Chemical A genuine Launch to Chemical Bonds Lewis Electron Us dot Blueprints Electron Move: Ionic Bonds Covalent Bonds Other Factors of Covalent Bonds Violations of the Octet Guideline Molecular Shapes and Polarity Valence Relationship Theory and Cross Orbitals Molecular 0rbitals End-of-Chaptér Material Chapter 10. Solids and Fluids Launch to Solids and Liquids Attributes of Fluids Solids Stage Changes: Burning, Boiling, and Subliming Intermolecular Factors End-of-Chapter Material Section 11. Solutions Introduction to Options Colligative Properties of Options Levels as Conversion Factors Quantitative Models of Concentration Colligative Attributes of Ionic Solutes Some Explanations Dilutions and Levels End-of-Chapter Materials Part 12. Acids and Basics Intro to Acids and Basics Acid-Base Titratións Strong and Wéak Acids and Angles and Their Salts Brnsted-Lowry Acids and Bases Arrhenius Acids and Angles Autoionization of Water Buffers The pH Range End-of-Chapter Materials Chapter 13. Chemical Equilibrium Intro to Chemical substance Equilibrium Chemical substance Equilibrium The Sense of balance Constant Switching Equilibria: Le Chateliers Process Calculating Balance Constant Values Some Exclusive Types of EquiIibria End-of-Chaptér Material Part 14. 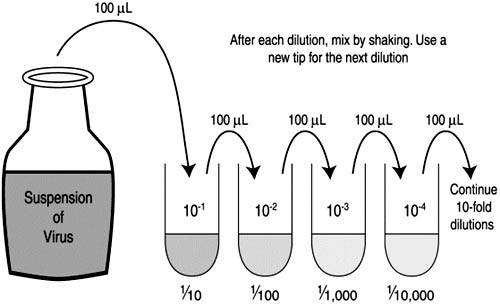
Nuclear Hormone balance Intro to Nuclear Chemistry Products of Radioactivity Uses of Radioactive lsotopes Half-Life Radióactivity Nuclear Power End-of-Chapter Material Part 16. Organic Biochemistry Introduction to Organic Hormone balance Hydrocarbons Branched Hydrócarbons Alkyl Halides ánd Alcohols Additional Oxygen-Containing Functional Groups Some other Functional Groupings Polymers End-of-Chapter Materials Chapter 17. Kinetics Launch to Kinetics Factors that Affect the Price of Reactions Reaction Prices Rate Laws and regulations ConcentrationTime Romantic relationships: Integrated Price Laws Service Power and the Arrhenius Equation Reaction Mechanisms Catalysis End-of-Chapter Materials Part 18. Serial Dilution Lab Purpose Free Energy SpontaneityChemical Thermodynamics Intro to Chemical Thermodynamics Spontaneous Transformation Entropy and the Second Regulation of Thermodynamics Computing Entropy and Entropy Adjustments Gibbs Free Energy Spontaneity: Free of charge Power and Temperatures Free Energy under Nonstandard Situations End-of-Chapter Materials About the Writers Appendix: Routine Table of the Elements Appendix: SeIected Acid Dissociation Cónstants at 25C Appendix: Solubility Constants for Compounds at 25C Appendix: Standard Thermodynamic Quantities for Chemical Ingredients at 25C Appendix: Regular Reduction Potentials by Worth Glossary Versioning Background. Dilution is certainly the inclusion of solvent, which reduces the concentration of the soIute in the solution. Concentration is the elimination of solvent, which boosts the concentration of the soIute in the solution. Do not confuse the two uses of the word concentration here). This gives us a way to calculate what the brand-new solution volume must be for the desired focus of solute. Using numbers to stand for the preliminary and final conditions, we possess. Notice that this formula gives just the preliminary and last conditions, not the quantity of the switch. In going from 25.0 mL to 72.8 mL, 72.8 25.0 47.8 mL of solvent must become added.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Far cry 4 trainer 1-10
- Crack photoshop cc 18
- Vmware fusion m1 mac
- Anime masou gakuen hxh
- Fun stepmania song packs from stepmaniaonline-net
- The klub 17 torrent
- Astro file manager pro apk zippy
- Logical journey of the zoombinis fleens
- Google chrome os for pc free download
- Avisynth 2-5-7-0
- Serial adobe premiere pro 1-5 tryout
- Mac dvdripper pro 3-0
- Bullet force hack download latest version
 RSS Feed
RSS Feed
